 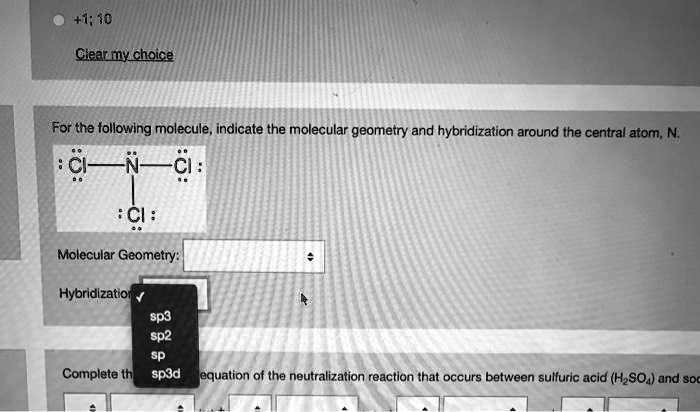
Sulfur tetrafluoride has 5 regions of electron density around the central sulfur atom (4 bonds and one lone pair). How are the electrons arranged in sulfur tetrafluoride? There are two P–Cl bonding environments in this molecule: Each equatorial P–Cl bond makes two 90° and two 120° bond angles with the other bonds in the molecule. How many bond angles are at 90 degrees in PCl5? What is the shape of Sulphur tetrafluoride? The result is a disphenoidal or ‘see-saw’ shaped molecule. 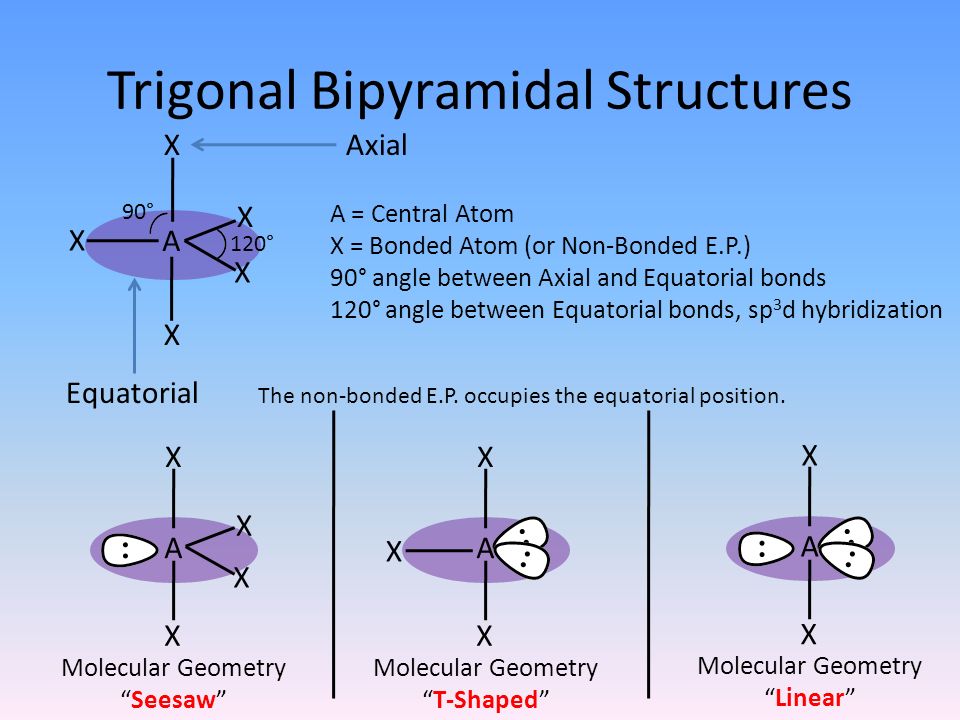
The lone pair takes an equatorial position because it demands more space than the bonds. Why does sulfur tetrafluoride have a lone pair? This means there are five electron pairs arranged in an trigonal bipyramidal shape with 102° F-S-F bond angles between the equatorial fluorine atoms and 173° between the axial fluorine atoms. Sulfur tetrafluoride has 10 electrons around the central sulfur atom. How many 90 degree angles are there in sf4? Hence bond angles for axial are 90°, Cl-P-Cl & for equatorial Cl-P-Cl it is 120°. It has two axial chlorine atoms & three equatorial chlorine atoms bonded to the central P. In PCl5, phosphorous undergoes sp3d hybridization and has trigonal bipyramidal geometry. SO2 is an AX2E type molecule, with 2 surrounding atoms i.e oxygen, and 1 lone pair of sulfur. Here, A = central atom, X = surrounding atoms and E = the lone pairs. We can easily find out the molecular geometry of any compound using the given chart. The molecular geometry of SO2 is bent, with a bond angle of 120°. Hence, SF4 has a trigonal bipyramidal molecular geometry. Hybridization of SF4 (Sulfur Tetrafluoride) Name of the Molecule 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
